AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Molar mass of carbon dioxide9/27/2023 
When all is ready pour 5–10 mL of the hydrochloric acid into the top of the thistle tube and allow it to run through the tube and into flask A. H2S co2 CH4 H20 Molar Mass, kg/kmol 34.082 44.010 16.043 18.015 Critical Temperature, K 373.5 304.2 190.6 647.1 Critical Temperature, C 100.4 31.1 -82.6.H h/M where: H is the specific enthalpy, kj/kg h is the molar enthalpy, kj/kmol (or J/mol) M is the molar mass of carbon dioxide, 44. The small rubber stopper attached to the bent glass tubing should be inserted into the other end of the drying tube. convert them to the specific property (per unit mass), divide by the molar mass of carbon dioxide (44.010 g/mol). Attach the flexible end of the tubing to the drying tube. Step 3: Use the molar mass of each fuel to find the mass of CO2 per gram of. Be careful to fold and shape the foil only as much as necessary since it is fragile and will easily tear. 5 All fossil fuels contain carbon and so release carbon dioxide when they. Insert the straight glass tube (the one that is attached to the flexible tubing but not attached to the one-holed rubber stopper) into flask B by placing it between the foil and the flask and pressing the foil against it to hold it in place. (Molar mass of CO2 44.0 g mo- (A) 1.00x 1024 (B) 6.80x1023 (C) 6.02x1023 (D) 3.40x 1023 This problem has been solved Youll get a detailed solution from a subject matter expert that helps you learn core concepts. Assemble the apparatus shown in figure 1.a pollution of 1 gram of benzene in a certain amount of water converts to N A/78.11≈ 7.\), in a small, labeled beaker. Using the above calculator you could find that e.g. Or 1 mole of a substance will contain Avogadro's number of that substance. One mole of carbon dioxide molecules has a mass of 44.01g 44.01 g, while one mole of sodium sulfide formula units has a mass of 78.04g 78.04 g. The molar mass of any compound is the mass in grams of one mole of that compound. The term " mole" is defined in that one mole of a substance with a molecular (or atomic) mass of one (1), will have a mass of 1 gram. The molecular mass of carbon dioxide is 44.01amu 44.01 amu. It is defined to be 1/12 of the mass of one atom of carbon-12 and in older works is also abbreviated as "amu".Īlso, important in this field is Avogadro's number (N A) or Avogadro's constant (6.0221 x 10 23). In related terms, another unit of mass often used is Dalton (Da) or unified atomic mass unit (u) when describing atomic masses and molecular masses. Molecular mass or molar mass are used in stoichiometry calculations in chemistry. This Calculator has been tested on Internet Explorer version 6 only,įirefox might not show all fields correctly. For question or remarks please contact us. !!! Lenntech BV cannot be held responsible for errors in the calculation, The molar mass of water (H2O) is 18.01 g/mol. 6CO2 + 6H2O -> C6H12O6 + 6O2 The molar mass of carbon dioxide (CO2) is 44.01 g/mol. Make sure you enter the molecule of crystallization at last (e.g. The chemical equation below shows the photosynthesis reaction.  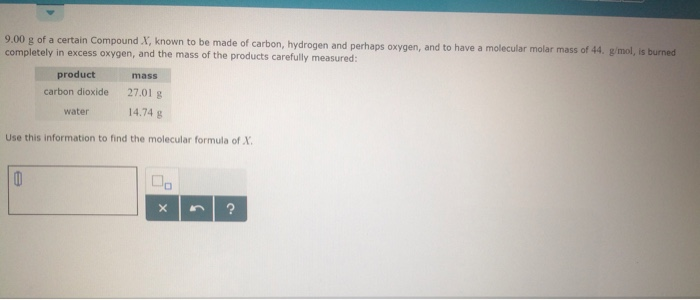
The calculator handles at most two different bracket levels. The molecular mass calculator will recognize the entered formula's, which are included in the list of organic compounds. Stoichiometric Equation Determine the moles of 'NaHCO'3' by dividing the given mass of 'NaHCO'3' by its molar mass, then determine moles of 'CO'2' by multiplying times the mole ratio with 'CO'2' in the numerator, then multiply times the molar mass of 'CO'2'. Or you can choose by one of the next two option-lists, which contains a series of common organic compounds (including their chemical formula) and all the elements. This online calculator you can use for computing the average molecular weight (MW) of molecules by entering the chemical formulas (for example C3H4OH(COOH)3 ). Molecular Weight Calculator Molecular Weight Calculator 
To compare the experimental molecular weight to the theoretical molecular weight of carbon dioxide.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
